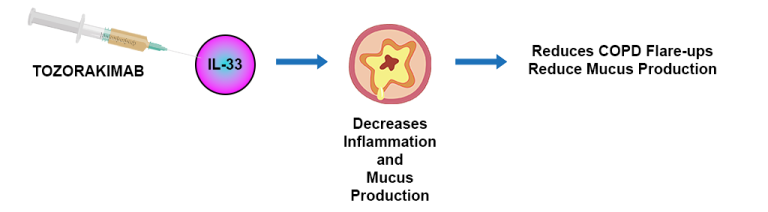
The study is designed to compare Tozorakimab with standard therapy to decrease the risk of flare-ups in COPD. It is an injected antibody that blocks IL-33, a protein that promotes inflammation and mucus production, thus leading to flare-ups, symptoms and disease progression in patients with COPD

If you participate, you will be randomly assigned to receive either injections of the study medication or placebo every 4 weeks. You will come for 15 visits and will be compensated $85 per clinic visit, and parking is provided. You will do questionnaires, lung function tests, daily home lung testing and maintain a daily electronic diary for symptoms. You may also have 2 CT scans, EKG, bloodwork and possibly 2 echocardiograms. You may have the opportunity to be part of a long term extension to continue with the medication.